Atrial Septal Defect (ASD)(Ostium Secundum Defect)
An atrial septal defect is one or more openings in the interatrial septum, producing a left-to-right shunt, pulmonary hypertension, and heart failure.
Symptoms and signs include exercise intolerance, dyspnea, fatigue, and atrial arrhythmias. A soft midsystolic murmur at the upper left sternal border is common.
 Diagnosis is by echocardiography. Treatment is surgical or catheter-based repair. Endocarditis prophylaxis is not usually required.
Diagnosis is by echocardiography. Treatment is surgical or catheter-based repair. Endocarditis prophylaxis is not usually required.
Atrial septal defects (ASDs) account for about 6 to 10% of cases of congenital heart disease. Most cases are isolated and sporadic, but some are part of a genetic syndrome (eg, mutations of chromosome 5, Holt-Oram syndrome).
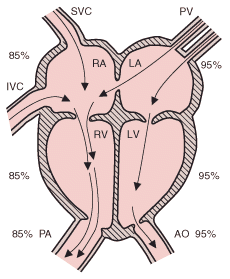
ASDs can be classified by location: ostium secundum (defect in the fossa ovalis—in the center [or middle] part of the atrial septum), sinus venosus (defect in the posterior aspect of the septum, near the superior vena cava or inferior vena cava), or ostium primum (defect in the anteroinferior aspect of the septum, a form of endocardial cushion defect (atrioventricular septal defect)).
In ASD, shunting is left to right initially. Most small ASDs close spontaneously during the 1st few years of life. However, persistent, large shunts result in right atrial and ventricular volume overload, pulmonary artery hypertension, elevated pulmonary vascular resistance, and right ventricular hypertrophy. Atrial fibrillation may occur later. Ultimately, the increase in right-sided pressure may result in a bidirectional atrial shunt with cyanosis during adulthood.
Symptoms and Signs
Most small ASDs are asymptomatic. Larger shunts may cause exercise intolerance, dyspnea during exertion, fatigue, and atrial arrhythmias sometimes with palpitations. Passage of microemboli from the venous circulation across the ASD (paradoxical embolization), often associated with arrhythmias, may lead to cerebral or systemic thromboembolic disorders. Rarely, when an ASD is undiagnosed or untreated, Eisenmenger's syndrome develops.
Auscultation typically reveals a grade 2 to 3/6 midsystolic (or ejection systolic) murmur and a widely split, fixed S2 at the upper left sternal border in children. A large left-to-right atrial shunt may produce a low-pitched diastolic murmur (due to increased tricuspid flow) at the left lower sternal border. These findings may be absent in infants, even those who have a large defect. A prominent right ventricular cardiac impulse may be present.
Diagnosis
Diagnosis is suggested by cardiac examination, chest x-rays, and ECG and confirmed by 2-dimensional echocardiography with color flow and Doppler studies.
With a significant shunt, ECG may show right axis deviation, right ventricular hypertrophy, or right bundle branch block (with rSR′ pattern in V1). Chest x-rays show cardiomegaly with dilation of the right atrium and right ventricle, a prominent main pulmonary artery segment, and increased pulmonary vascular markings.
Cardiac catheterization is not usually necessary unless additional associated anomalies are suspected.
Treatment
Most small ostium secundum ASDs (< 3 mm) close spontaneously; about 80% of those between 3 mm and 8 mm close spontaneously by age 18 mo. However, ostium primum and sinus venosus ASDs do not close spontaneously.
Asymptomatic children with a small shunt require annual echocardiography. Because these children are at risk of paradoxical systemic embolization, some centers recommend a catheter-delivered closure device (eg, Amplatzer Septal Occluder, Cardio-Seal device) even for small ASDs. However, these devices are not suitable for primum or sinus venosus defects because these defects are near important structures.
Patients with moderate to large defects (eg, pulmonary to systemic flow ratio > 1.5:1) should have the ASD closed, typically between age 2 to 6 yr. Closure devices are preferred when appropriate anatomic characteristics are present and the defect is < 13 mm in diameter. Otherwise, surgical repair is indicated. If ASDs are repaired during childhood, perioperative mortality rate approaches 0, and long-term survival rates approach those of the general population.
http://www.merck.com/mmpe/sec19/ch287/ch287b.html



